
todamo/iStock Editorial via Getty Images
Mirati’s Success With PRMT5 Inhibitor Puts Spotlight On Tango
Back in October last year I posted a note on Seeking Alpha related to Bristol-Myers Squibb (BMY) and its decision to complete the acquisition of Mirati Therapeutics (MRTX) in a deal worth $58 per share, for a total equity value of $4.8bn. The deal completed this week, with Mirati becoming a wholly owned subsidiary of Bristol Myers Squibb.
Mirati – which has been in the news this week for other, more unexpected reasons, named as one of the companies being traded in an alleged insider trading case involving British billionaire investor Joe Lewis – is a drug developer focused on oncology that secured approval for a drug, Krazati, targeting the Kirsten rat sarcoma virus, or KRAS, in late 2022 – a feat widely admired as KRAS, found be mutated in a host of solid tumor cancers, had long been considered undruggable.
BMY’s deal for Mirati could end up rising to $5.8bn in value, based on the progress of a second drug candidate developed by Mirati, known as MRTX1719. According to BMY’s press release announcing the deal last October:
Each Mirati stockholder will also receive one non-tradeable CVR per Mirati share, which will entitle its holder to receive a one-time potential payment of $12.00 in cash, for a total value of approximately $1.0 billion, upon acceptance by U.S. FDA of a new drug application for MRTX1719 for the treatment of either locally advanced or metastatic NSCLC in patients who have received no more than two prior lines of systemic therapy within seven years after the closing of the merger, subject to the terms and conditions contained in a contingent value rights agreement detailing the terms of the CVR.
On Mirati Therapeutics’ website, MRTX1719 is discussed as follows:
MRTX1719 is an investigational, internally discovered MTA-cooperative PRMT5 inhibitor that selectively binds the PRMT5-MTA complex, inhibiting PRMT5 function in MTAP-deleted cancer cells. MRTX1719 selectively inhibits the viability of cancer cells with MTAP deletion and is designed to spare healthy, non-tumor cells.
Methylthioadenosine Phosphorylase or MTAP is a gene that frequently suffers homozygous deletion in cancer which results in the accumulation of methylthioadenosine (“MTA”) within cancer cells. MTA binds to PRMT5 and forms the PRMT5-MTA complex. This new complex creates a novel drug target for the potential treatment of MTAP-deleted cancers.
Research suggests ~10-15% of all cancers are MTAP-deleted, suggesting that a successful PRMT5-inhibitor drug could target a substantial market opportunity – clearly, BMY’s deal for Mirati was not just about gaining access to Krazati.
This brings me to the topic of this post – a Boston-based biotech named Tango Therapeutics (NASDAQ:TNGX). Besides Mirati, and the Californian Pharma giant Amgen (AMGN) (which also developed the only other approved KRAS targeting drug besides Krazati to have been conditionally approved by the FDA, Lumakras, although it has since been refused a full approval), Tango is the only other company with a clinical stage, MTA-cooperative PRMT5 inhibitor drug in development.
In fact, Tango has not one, but two PRMT5 inhibitors in clinical development – TNG908 is its first-generation candidate, and TNG462 its next generation candidate, and both are enrolling patients in a Phase 1/2 study. Tango listed on the Nasdaq via a deal with a Special Purpose Acquisition Company (“SPAC”), Boxer Capital’s BCTG Acquisition Corp (also believed to be one of the four companies named in the case against Joe Lewis, mentioned above).
Tango Shares Spike On Promising Mirati Data For PRMT5-Inhibition – Moves Candidates Into Clinic
Since its listing in August 2021, Tango’s share price performance has been somewhat volatile, with the stock mainly trending downwards, reaching a low of ~$3 in July last year, before surging to a value of ~$11.5 by mid October, buoyed by encouraging data released by Mirati in relation to MRTX1719.
Mirati highlighted six confirmed objective responses in a study of 18 patients with solid tumors harboring MTAP deletions, and “achieved apparent complete PRMT5 inhibition in MTAP deleted tumor cells at a dose of 200 mg QD,” according to a press release issued at the time. Encouragingly, the press release also noted:
- MRTX1719 was well-tolerated with no dose limiting toxicities observed at dose levels up to 400mg QD. None of the patients treated with MRTX1719 experienced dose-limiting adverse events associated with first generation PRMT5 inhibitors such as thrombocytopenia, anemia or neutropenia.
Mirati also observed that the data indicated “tumor response may continue to deepen over time.” In short, the data appeared to provide some early validation for the PRMT5 inhibitor drug class, and as one of only two other companies with such a drug in the clinic, Tango’s shares spiked in value.
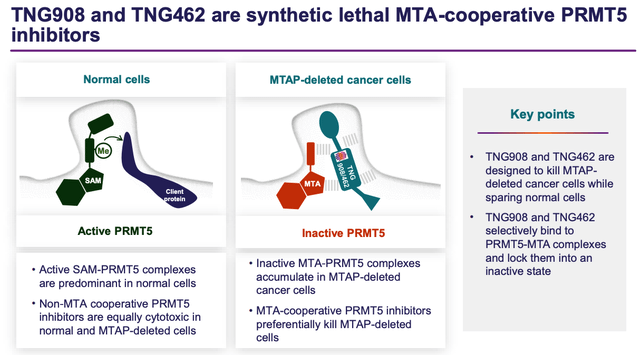
TNG908 / TNG462 mechanisms of action (Tango investor presentation)
As we can see above (from a slide taken from a recent Tango presentation), the mechanism of action of Tango’s two PRMT5-inhibitors is founded upon another innovative new approach known as synthetic lethality, described as follows in Tango’s 2022 annual report / 10K submission:
Synthetic lethal therapies for cancer refers to pairs of genes where one is inactivated by a genetic alteration and the other is inhibited pharmacologically.
While genetic alterations give rise to the development of cancer, they also create a unique vulnerability that can be exploited therapeutically. Biologically, such vulnerability can be the inability of cancer cells to respond to a specific signal, such as DNA damage or cell cycle arrest, or the inability to remodel chromatin or to maintain cellular homeostasis.
The unique advantage of a synthetic lethal approach to cancer therapy is that normal cells are not vulnerable to the synthetic lethal drug target and are largely unaffected at drug doses where the mutant cancer cells are selectively killed.
One of the major difficulties involved in treating cancers is how to avoid damaging healthy cells when targeting cancer cells – synthetic lethality may potentially provide a solution to this problem that, while not perfect, may be an improvement on current, commercially available, standards of care.
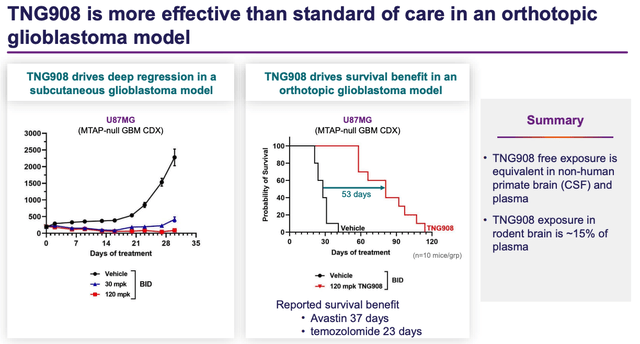
TNG908 in GBM model (Tango presentation)
As shown above, in a preclinical study in animal models, TNG908 exhibited a potentially best in class survival benefit, comfortably outperforming the likes of Avastin – marketed and sold by Swiss pharma giant Roche (OTCQX:RHHBY), and earnings >$2bn of revenues in 2022, and US pharma Merck’s (MRK) Temodar.
Similarly, TNG462’s preclinical data marks it out as a “potentially best-in-class” PRMT5 inhibitor, Tango believes, based on tumor growth inhibition in 22/22 xenograft models, with tumor regression observed in 55% of models (versus 30% for TNG908).
In summary, Tango appears to have developed two potentially exciting candidates in an innovative new class of drugs exhibiting strong preclinical data. With that said, the company must still prove its drugs can be effective in the clinic, likely through Phase 1, 2 and 3 studies, before a push for approval can even be contemplated, and that will take time, resources, and plenty of cash.
As mentioned, Phase 1/2 studies have been initiated for both candidates, and there has been some early encouragement – according to Tango’s Q3 10Q submission / quarterly report:
Patients are actively being enrolled in the Phase 1/2 clinical trial. Initial pharmacodynamic (“PD”) data from the ongoing TNG908 dose escalation study, released in May 2023, provided proof-of-mechanism of MTA-cooperative PRMT5 inhibition, demonstrated by marked reduction of SDMA staining in MTAP-deleted cancer cells vs. normal tissue.
Pre-treatment and on-treatment biopsies demonstrated dose-dependent decreases in tumor SDMA staining with minimal or no decrease in normal tissue. The selective inhibition of PRMT5 in MTAP-deleted cancer cells is essential to enable the therapeutic index needed for efficacy. Additional TNG908 clinical data are expected in 2024.
TNG908’s study involves a dose expansion cohort which will cover a number of solid tumor cancers, including non-small cell lung cancer – one of the largest oncology markets – glioblastoma, pancreatic, sarcoma, and mesothelioma – the SDMA reduction mentioned above occurred in an ovarian cancer patient.
The update due this year may include some data around actual patient responses e.g. partial, objective, or complete, although management has not promised any data from the TNG462 study as yet. It should be noted that this candidate is not brain penetrant, meaning it will not be evaluated in GBM.
Tango’s Two Other Candidates Hedge Risk, Optimize Innovation
While “Synthetic Lethality” and “PRMT5” are rapidly becoming buzzwords that generate a positive response from the market, there’s no question that there’s a long and tortuous path for developers to pursue before we may see such a drug formally approved to treat patients in a commercial setting, and therefore this may be a good time to remind ourselves that cancer drugs are estimated to have a less than one in twenty chance of successfully making it from the Phase 1 stage, all the way through approval.
As such, it is comforting (although it by no means completely derisks an investment in the company) that Tango has 2 other promising candidates in its pipeline.
The first, TNG260 is “a highly selective CoREST complex inhibitor – according to Tango, the drug:
reverses the immune evasion effect of STK11 loss-of-function mutations. STK11 loss-of-function mutations are present in approximately 15% of NSCLC, 15% of cervical cancers, 10% of carcinoma of unknown primary, 5% of breast cancers and 3% of pancreatic cancers.
TNG260 is designed to work alongside an anti PD (programmed death)-1 inhibitor – for example, Merck’s all-conquering cancer mega-blockbuster keytruda, or Bristol-Myers Squibb’s (BMY) Opdivo (>$8bn of revenues in 2022).
Tango says that “TNG260 in combination with an anti-PD-1 antibody is active in cancers with STK11 deletion, a setting where an anti-PD-1 antibody alone is inactive,” which, if true, would open up new market opportunities for PD-1s and therefore, significant new market opportunities for TNG260.
The Investigational New Drug (“IND”) application for the candidate has been cleared by the FDA, allowing in-human studies to begin, and a first patient was dosed in a clinical study, alongside keytruda, in July last year, in solid tumor cancers.
And finally, Tango’s fourth candidate, TNG348, is focused on USP1 inhibition, a mechanism of action explained in the slide below (again taken from a recent Tango presentation).
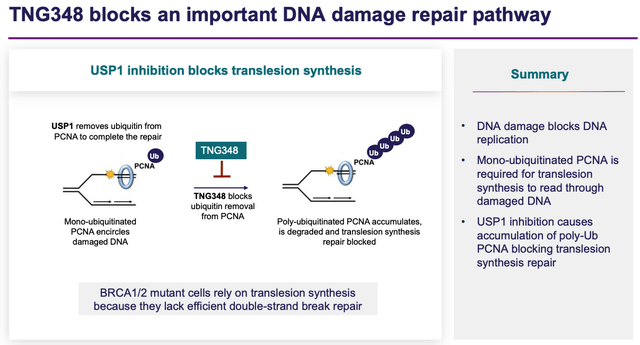
USP1 inhibition explained (investor presentation)
TNG348 will target BRCA1, BRCA2-mutant and other HRD+ cancers, which Tango says “represent approximately 50% of ovarian, 25% of breast, 10% of prostate and 5% of pancreatic cancers.”
Intriguingly, Tango intends to partner this candidate with yet another “bleeding edge” cancer treatment approach, in the form of a PARP inhibitor. PARP, which stands for “poly-ADP ribose polymerase,” helps damaged cells repair themselves, but its inhibition can prevent cancer cells from doing so, helping shrink tumors.
Three drugs with this MoA have been approved to date, AstraZeneca’s (AZN) Lynparza ($2.6bn revenues in 2022) and GSK’s (GSK) Zejula ($463m of revenues in 2022), both indicated for ovarian cancer, and the now bankrupt and delisted Clovis Oncology’s Rubraca, which has been acquired by Swiss company Pharma& Schweiz GmbH. The drug earned $164m of revenues in 2020. TNG348’s IND has been cleared, and a clinical study is expected to begin this year.
Concluding Thoughts: Tango Will Divide Opinion But I See Long-Term Promise
Biotech investing is a risky business and it’s rare to come across any listed biotech that is not waving a red flag or two in the direction of prospective investors.
In the case of Tango, we can start with the fact that its recent gains are in fact based on the performance of another company – Mirati Therapeutics’ – drug candidates in the clinic, and not its own. It is Mirati’s MRTX1719 that has generated objective responses in the clinic, not Tango’s TNG462, or TNG908. Therefore, we could argue Tango shares recent rise in value was not justified.
It may be additionally worth noting that Mirati’s data is from a Phase 1/2 study only, and perhaps we should also consider the fact that the jury is still very much out on Mirati’s approved candidate, Krazati, and whether it will prove a commercial success.
As I mentioned earlier, Amgen’s KRAS targeting asset Lumakras risks being permanently withdrawn from the market, indicating that not all promising new drug targets become viable commercial opportunities, and it’s certainly noteworthy that Amgen’s data for its own PRMT5 inhibitor were a disappointment, with only five of 31 patients achieving partial responses, underlining the difficulty of turning a promising a new approach into an effective drug, no matter how positive the preclinical data.
Meanwhile, AstraZeneca (AZN) has emerged as another competitor in the PRMT5 space, moving its next generation candidate AZN3470 into the clinic, and leaving former partner Schrödinger to develop its own candidate under its own stream. In short, the competition is heating up.
At present, Tango’s losses – $(79.2m) across the first 9m of 2023 – vs. near-term cash of ~$370m, reported as of Q3, suggest the company has a cash runway into 2026, and management has confirmed that it expects to achieve Proof-of-Concept (“PoC”) readouts for all four of its clinical programs before its cash runs out. Ultimately, however, can Tango take on the might of big pharma companies like AstraZeneca and Bristol Myers Squibb?
When a new type of drug exhibits exciting new potential in the clinic, it’s almost inevitable that a handful of biotechs at least will switch focus to the new drug class, or raise money to develop drugs in that class, and as such it can be difficult to determine which companies are the “real deal,” and which are attempting to position “copycat” drugs as potential new standards of care.
From a layman’s perspective, it’s far from easy to separate the “wheat from the chaff” – witness the sheer number of companies that pivoted into COVID vaccine development during the pandemic, enjoying staggering rises in the share price, and ultimately failed – nevertheless I am going to stick my neck out and back Tango for some success.
It’s often said “do not invest in what you don’t understand” – a mantra that might put most investors off the pharma / biotech industry altogether – and it is tempting to wonder how many biotechs might prefer investors not to know exactly how their drugs work, as they may not know themselves.
In the case of Tango, however, I personally find the company’s approach based on concepts that appear logical and based on sound scientific research. We know that there are a high number of patients with MTAP deleted cancers, for example, we know that synthetic lethality may represent another step forward in the fight to avoid “off target toxicity” (damaging healthy cells), and we have some quite persuasive preclinical data to show some early evidence of proof-of-concept.
We have a potentially important data catalyst arriving this year for lead candidate TNG908, which is brain penetrant, while the explanation for developing a next-generation candidate on the heels of TNG908 is plausible, in my view.
Unlike so many early stage biotechs, Tango is not a single-asset company, trumpeting a “pipeline-in-a-pill” drug candidate. There’s good diversification to be found in TNG260 and TNG348, and I also find it encouraging that these drugs have been partially developed to work alongside other, more established drug classes, such as PARP inhibitors and immune checkpoint inhibitors (“ICIs”). Again, I find the explanation, particularly around uncovering new opportunities for ICI’s such as keytruda / opdivo, plausible, and likely to attract the attention of Big Pharma partners.
Tango is still further derisked by a partnership with Gilead Sciences (GILD), a leading big pharma led by an oncology veteran, attempting to pivot more and more towards treating cancer patients. The partnership will be based on the discovery and development of up to 15 targeted immune evasion targets. Gilead has made an upfront payment of $125m to Tango, with Tango, according to its Q3 2023 10Q:
… eligible to receive up to $410.0 million per program in license, research option-extension, and clinical, regulatory, and commercial milestones and royalties on future sales of commercialized products.
Unfortunately, there’s no way for a clinical stage biotech to ever become a fully derisked investment opportunity, and another drawback I note in relation to Tango is the lack of near-term catalysts, with only one data readout of note likely to occur this year, in a Phase 1/2 study.
Bearing that in mind, Tango stock may feel expensive with the company’s current valuation of $1.3bn, an order of magnitude higher than most biotechs which have only just entered the clinic.
Nevertheless, I feel a higher price may potentially be worth paying. While antibody drug conjugates may have been the biotech / oncology story of last year, with Pfizer (PFE) shelling out $43bn to acquire Seagen, and AbbVie (ABBV) ~$10bn to acquire Immunogen (IMGN), and their approved ADC candidates, it may be the case that synthetic lethality – which follows a similar logic to ADC development – could become the story, if not of 2024, then perhaps 2025. The year does not matter essentially, so long as the data exhibits best-in-class potential.
There may be opportunities to buy Tango stock at much cheaper prices that today’s price of $13 per share, as the stock price is clearly volatile and influenced by the progress of other drug developers, rightly or wrongly. Inevitably, however, if Tango is on the right track, its valuation ought to soar – at one stage, Mirati stock was worth >$250 per share, and the business valued >$15bn.
I would urge prospective investors to be patient with Tango, however, and let the four clinical studies progress. With no dilution likely unless candidates return positive, share price needle moving data, I like Tango as a long-term admittedly high risk, biotech play.



