PeopleImages/iStock via Getty Images
At a Glance
On Tuesday, Sanofi (NASDAQ:SNY) announced it had acquired Inhibrx’s (NASDAQ:INBX) lead rare disease asset, INBRX-101, for up to $2.2 billion. The deal is just one of a few deals Sanofi has made in an attempt to broaden their pipeline, specifically for rare diseases. My last update on Sanofi expressed concerns regarding the spin-off of their Consumer Healthcare segment, making them, in my view, more vulnerable to obvious deficiencies in drug development and retention (e.g., Lantus). In addition to discussing Sanofi’s metrics in comparison to its peers and its ability to fund the acquisition, the piece that follows explains the implications of the INBRX-101 acquisition for Sanofi and its shareholders.
Sanofi Springs for Rare Cure: INBRX-101’s $2.2 Billion Gamble
Sanofi’s bold leap, acquiring Inhibrx’s INBRX-101, targets rare diseases. This aligns with its strategy. INBRX-101, a therapeutic for Alpha-1 Antitrypsin Deficiency (AATD), holds promise. AATD is a genetic disease affecting the lungs and liver.
INBRX-101’s design is novel. It’s an Alpha-1 Antitrypsin (AAT)-Fc fusion protein. The goal is to normalize serum AAT levels. It aims for less frequent dosing. Existing treatments require more regular administration. INBRX-101’s monthly schedule, as opposed to weekly, stands out. This could improve upon the existing AATD treatment. Importantly, INBRX-101’s approach is validated, as current treatments focus on repleting Alpha-1 protein via weekly infusions.
For Sanofi, this is a continuation of their strategic expansion into rare diseases. Previous efforts include a proposed $150 million Maze deal for Pompe disease in May 2023. However, this deal was later struck down by the FTC, citing Sanofi’s existing “monopoly” in Pompe disease. Moreover, since 2018, their Rare Blood Disorders franchise has been growing. Acquisitions and in-house programs played a role. For example, the $3.6 billion Principia buyout in 2020 expanded their immune-mediated blood disorder work.
They’ve also established Rare Disease Registries. These collect real-world data on diseases like Gaucher, Fabry, MPS I, and Pompe. This data is crucial for advancing research and treatment for rare diseases.
Financial Health
Upon glancing at Seeking Alpha data, Sanofi’s financial position as of September 30, 2023, appears robust, particularly in the context of their ability to fund the $2.2 billion acquisition of Inhibrx’s INBRX-101. Their cash and short-term investments, amounting to $8.45 billion, underscore a strong strategic investment capability. Receivables stand at an impressive $9.43 billion, reinforcing liquidity. The total current assets, valued at $33 billion, further solidify their financial strength. Long-term assets, including property, plant, equipment, and intangibles, contribute to a significant asset base, while total liabilities of $57.18 billion are balanced against substantial assets and equity. Notably, their total common equity is high at $76.78 billion.
Sanofi’s debt situation is also a critical aspect, with total debt at $22.23 billion and a net debt of $13.78 billion. This level of indebtedness is a key factor in their financial capability to manage such a significant acquisition.
Overall, Sanofi’s balance sheet reveals a solid financial standing, with ample cash reserves, high assets, and robust equity, suggesting their capacity to finance the INBRX-101 acquisition without jeopardizing financial stability.
Peer Comparison
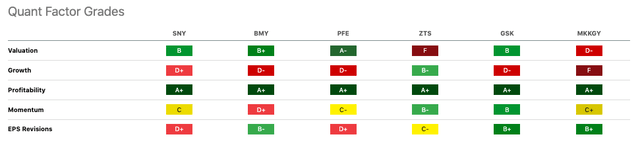
Sanofi presents a unique juxtaposition in the pharmaceutical landscape. It differs distinctly from Bristol Myers Squibb (BMY), Pfizer (PFE), Zoetis (ZTS), GSK (GSK), and Merck (OTCPK:MKKGY).
In valuation terms, Sanofi’s attractiveness shines. It boasts a P/E Non-GAAP (FY1) of 11.40 and a P/E GAAP [TTM] of 13.43. These figures position it favorably against Pfizer and Zoetis. However, it lags behind BMY and GSK. Its moderate price-to-sales and EV-to-EBITDA ratios hint at investment-worthy potential.
Growth metrics for Sanofi show a mixed bag. A modest 3.05% revenue growth (YoY) stands out, especially against Pfizer’s steep -31.38% dip. Over three years, its revenue CAGR of 6.67% demonstrates consistent expansion. Yet challenges loom in earnings growth, as seen in diluted EPS metrics.
Profitability is Sanofi’s strength, with an exemplary A+ rating. Its gross profit, EBIT, and net income margins are robust, though slightly trailing peers like BMY and ZTS. Returns on equity and assets are commendable, albeit not the top in their category.
Dividend-wise, Sanofi excels with a forward yield of 3.74%. This rate surpasses ZTS and MKKGY but falls short of BMY and PFE. Its dividends are stable, though not remarkably so.
To conclude, Sanofi’s profile is multifaceted. Valuation, consistent growth, and profitability are its strengths. Its dividend appeal is clear, offering a blend of growth and income opportunities in healthcare. However, EPS growth needs bolstering. Sanofi recognizes this and makes strategic acquisitions such as INBRX-101 to enhance their growth chances.
Is SNY Stock a Buy, Sell, or Hold?
Overall, I like this deal for Sanofi. It makes sense. INBRX-101 fits well within their expertise and doesn’t come with a hefty price tag. Although INBRX-101 is still in development, because its approach is validated, it appears to have a simple and easy path to the market. Upon entry, INBRX-101 has been estimated to pull upwards of $623 million in peak annual revenue. Given that INBRX-101 is a biologic and treats a rare disease, it should have a favorable exclusivity period.
Sanofi’s stock is still a “hold,” in my opinion; while this is a step in the right direction, revamping drug pipelines is a difficult task, and Sanofi still has much to prove. Nonetheless, Sanofi could be an interesting addition to any diversified portfolio. Investors should keep an eye on quarterly financial performance and pipeline updates. Also, be aware of the risks associated with investing in general. In this case, it’s possible that INBRX-101 fails in the clinic, and Sanofi is left with empty pockets, shrugged shoulders, and palms facing the sky. Until that happens, I believe it is safe to presume Sanofi has another solid rare disease asset in their possession at a reasonable cost.